Background
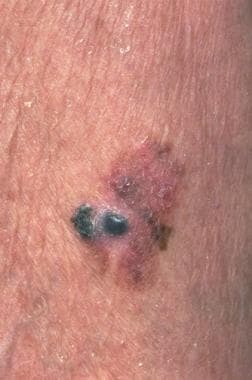
Melanoma is a malignancy of pigment-producing cells (melanocytes), which are located predominantly in the skin but also found in the eyes, ears, gastrointestinal tract, leptomeninges, and oral and genital mucous membranes. Characteristic images of cutaneous melanoma are shown below.
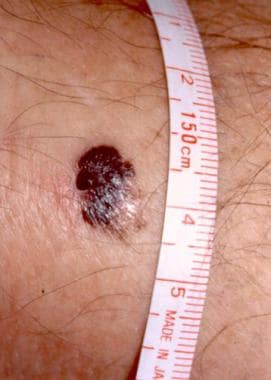 Cutaneous melanoma with characteristic asymmetry, irregular borders, and color variation. Courtesy of Wendy Brick, MD.
Cutaneous melanoma with characteristic asymmetry, irregular borders, and color variation. Courtesy of Wendy Brick, MD.
Invasive melanoma accounts for only 1% of all skin cancers in the United States but causes the greatest number of skin cancer–related deaths. [1] Early detection of thin cutaneous melanoma is the best means of reducing mortality, although mortality rates have dropped over the past decade due to the advent of more effective targeted and immune therapies for patients with advanced disease. [2]

Pathophysiology
The transformation of normal melanocytes into melanoma cells, referred to as melanomagenesis, is believed to involve a multistep process of progressive genetic mutations. Along with genetic damage from ultraviolet radiation (UVR), the development of melanoma has been linked to specific genetic alterations that attenuate mechanisms of tumor suppression or activate signalling pathways that promote growth. [3] These mutations (1) alter cell proliferation, differentiation, and death and (2) impact susceptibility to the carcinogenic effects of UVR. [4]
Genome-wide analysis of these genetic aberrations has elucidated the complex interplay of signaling pathways that lead to melanoma pathogenesis, [5] as well as the contributions of phenotypic and environmental factors. For instance, melanomas on sun-protected skin (trunk) tend to develop in association with a high nevus count and intermittent UVR exposure, whereas those developing on sun-exposed skin are associated with low nevus count and chronic UVR exposure. [6, 7]
Differences in frequency of BRAF or NRAS oncogenic mutations are also related to patterns of sun exposure, with BRAF mutations more common in intermittently UVR-exposed skin compared with chronically sun exposed skin or relatively unexposed skin (eg, acral sites, mucosal sites), which more frequently demonstrate KIT mutations. [7, 8] A meta-analysis by Lee et al demonstrated that the prevalence of these mutations may also depend on melanoma histologic subtype. [9]
Primary cutaneous melanoma may develop in association with precursor melanocytic nevi (ie, common, congenital, and atypical/dysplastic types), although more than 70% of cases are believed to arise de novo (ie, not from a preexisting melanocytic neoplasm). Some data suggest that de novo melanomas have a more aggressive biology (ie, likely to be thicker, ulcerated, and later stage) than those that are nevus associated. [10]
The development of melanoma appears to be related to multiple risk factors, including the following [11, 12, 13] :
-
Lighter skin complexion/sun sensitivity
-
Excessive childhood sun exposure and blistering childhood sunburns
-
An increased number of common or atypical/dysplastic nevi (moles)
-
Family history of melanoma
-
Older age

Etiology
Worldwide, the incidence rate of melanoma is highest in lighter-complexioned individuals living in sunny climates and nearer the equator, supporting a causative role for ultraviolet radiation (UVR). Most data support the hypothesis that melanoma development is linked to intermittent, intense sun exposure beginning in childhood or adolescence over chronic sun exposure, [14, 15] with the exception of the more UVR-related melanoma subtypes (lentigo maligna and invasive lentigo maligna melanoma). The use of tanning beds (artificial UVR) has also increased the incidence of melanoma, most notably in younger patients. [16]
Primary risk factors and clinical warning signs for melanoma include the following:
-
Changing mole (most important clinical warning sign)
-
Presence of xeroderma pigmentosum or familial atypical mole melanoma syndrome: These genodermatoses confer a 500- to 1000-fold greater relative risk of developing melanoma
-
Clinical atypical/dysplastic nevi in familial melanoma
-
Sporadic (nonfamilial) clinical atypical/dysplastic nevi (particularly > 5-10)
-
Melanoma in one or more first-degree relatives (especially multiple)
-
Large numbers of common/typical nevi (> 100)
-
Previous melanoma
-
Male sex
-
Age older than 50 years
-
Sun sensitivity/history of excessive sun exposure or sunburns
-
Large (giant) congenital nevi (> 20 cm diameter in an adult)
-
Prior nonmelanoma skin cancer (ie, keratinocyte carcinomas: basal cell and squamous cell carcinoma)
-
Immunosuppression
Lighter-skin phenotype (blue/green eyes, blond or red hair, light complexion, sun sensitivity) and the experience of blistering sunburn(s) in childhood and adolescence are universal risk factors for melanoma. Individuals with these factors have been the focus of preventive efforts worldwide, although a growing burden of melanoma has been observed in individuals with darker skin. [17, 18, 19]
Long-term treatment with certain immunosuppressive drugs may increase risk of skin cancer, but the risk is principally for non-melanoma skin cancer (eg, squamous cell carcinoma, basal cell carcinoma). Regular screening for skin cancer is recommended for solid organ transplant recipients and for select nontransplant patients who are receiving immunosuppressive regimens associated with increased risk. [20, 21]
There is contradictory evidence on the possibility of increased melanoma risk with use of exogenous female sex hormones (ie, oral contraceptives, hormone replacement therapy), and during pregnancy, perhaps from photosensitizing effects of estrogen. However, any elevation in risk is at most borderline. [22, 23, 24, 25] American Academy of Dermatology melanoma clinical practice guidelines state that women who have had cutaneous melanoma may use exogenous hormones, and do not need a prolonged waiting period before subsequent pregnancy, given the lack of evidence that exogenous hormones or subsequent pregnancy affect prognosis in these women. [26]
Certain occupations are associated with increased risk for melanoma. Firefighters and military personnel are at increased risk due to exposure to potential carcinogens in combustible by products and other chemicals, likely compounded by UVR. [27, 28, 29, 30] Airline pilots and flight crews demonstrate melanoma risk double that of the general population. This can be attributed to higher exposure to cosmic radiation and UVR. [31]

Epidemiology
Frequency
United States
The incidence of melanoma has more than doubled in the White population over the last 30 years. Melanoma currently is the fifth most common cancer in the United States in both men and women. [1] Surveillance, Epidemiology, and End Results (SEER) data from 1975 to 2017 showed continued increases in melanoma incidence at all anatomic sites, except for head and neck melanomas in men, although much of this increase was driven by detection of thin tumors (< 1.5 mm).
The American Cancer Society estimates that 104,960 people in the US (60,550 men and 44,410 women) will develop invasive cutaneous melanoma in 2025, with an estimated additional 107,240 or more cases of melanoma in situ. [1] The actual incidence of melanoma may be higher due to underreporting of melanoma to cancer registries, particularly for in situ and thinner tumors that are diagnosed and managed in the outpatient setting. [32]
On the other hand, a study of SEER data from 1975 to 2014 found discrepancies in incidence and mortality trends among Black and White patients that suggested melanoma overdiagnosis of as much as 60% in White patients. In Blacks during that period, the melanoma incidence increased by less than 25% and mortality decreased by approximately 25%. In Whites, the reported incidence rose approximately 4-fold in men and 6-fold in women. Had trends been comparable to those in Blacks, mortality in Whites would have increased 60% in women and more than doubled in men; instead, mortality was stable in women and increased by less than 50% in men. [33]
Rates of melanoma in situ (intraepithelial) have also steadily risen to equal those of invasive melanoma, raising concerns regarding overdiagnosis of melanocytic neoplasms that would otherwise prove harmless. [34] Those concerns have prompted international expert pathology efforts to explore the impact that alternative diagnostic labels (eg, "low-risk melanocytic neoplasm" rather than "melanoma in situ") might have on patients' choice of treatment and their anxiety levels. [35]
Encouragingly, decreasing melanoma incidence rates have been noted in younger age groups (below age 50 years) in the United States, [36] which may be a result of primary prevention campaigns over the past 30 or more years aimed at reducing excessive sun exposure. However, the full impact of public health strategies on melanoma incidence will not be apparent for some time to come.
Melanoma incidence more than doubled from 1980-2004 in White women younger than 40 years. The rise is attributed at least in part to increased UVR exposure through tanning bed use, which is a World Health Organization (WHO)–classified carcinogen. [37]
A study assessing melanoma incidence among younger White girls and women (15-39 y) in California showed that in areas with the highest UVR exposure, the incidence of melanoma was significantly increased in higher than in lower socioeconomic neighborhoods, suggesting that affluence (and associated lifestyle behaviors) could have a greater impact on melanoma risk than outdoor UVR exposure alone. [38] These trends prompted public health and legislative efforts to prohibit indoor tanning bed use in minors and to ban indoor tanning entirely in the US, as has been done in several other countries.
International
Melanoma incidence has continued to increase worldwide. Globally, melanoma was the 17th most common cancer in 2022. [39]
Australia and New Zealand have the highest rates of melanoma. Analysis of global cancer statistics for melanoma from 2022 demonstrated an age-standardized incidence rate of 35.8 cases per 100,000 men and women in Australia and New Zealand, compared with 18.0 in northern Europe and 16.3 in northern America. [39] However, melanoma incidence in Australia has decreased by 5% in individuals age 15-24 years, likely reflecting the success of skin cancer prevention campaigns. [40]
Race
In the United States, the current lifetime risk of developing melanoma is about 2.6% (1 in 38) for Whites, 0.6% (1 in 167) for Hispanics, and 0.1% (1 in 1000) for Blacks. [41] Although melanoma is primarily a malignancy of White individuals, mortality rates are higher in Blacks and Hispanics, who are more likely to have acral melanoma and later-stage disease at presentation. [42]
Sex
In the United States, invasive melanoma has a higher female predilection from birth to age 49 years. However, from age 50 years on, melanoma in men predominates. [1]
Worldwide, of the 331,722 new cases estimated to have occurred in 2022, men were affected slightly more than women, with 179,953 versus 151,769 cases, respectively. Of the estimated 58,667 worldwide deaths in 2022, 33,160 were in men and 25,507 in women. [39]
Age
The median age at melanoma diagnosis is 66 years. [41] However, melanoma is the most common cancer in women aged 25-29 years and is second only to breast cancer in women aged 30-34 years. [43]
From 2010 through 2014, melanoma incidence decreased slightly in younger non-Hispanic White men and women but continued to increase significantly in men older than 54 years and women older than 44 years. [44] The most striking differences in melanoma incidence and mortality occur in individuals older than 65 years, although modest differences in age-specific incidence and mortality are notable in persons older than 50 years. [45]
Older individuals are both more likely to acquire and to die from melanoma (particularly White men aged 65 years and older), marking them a primary target for early detection and screening. [46] Treatment options in elderly persons may also be limited because of comorbid medical conditions, an inability to tolerate adverse medication effects or toxicity, the increased likelihood of drug interactions, and potential exclusion from clinical trials based on age criteria, although newer immune and targeted therapies are often well tolerated patients of advance age. [46]

Prognosis
Tumor stage at diagnosis has greater importance for the prognosis in melanoma than in almost any other cancer. [3] Despite remarkable advances in the treatment of metastatic disease, detection and treatment of cutaneous melanoma in its thinner, early phase remains the best chance for cure.
Prognosis is multifactorial and primarily depends on the following factors at initial diagnosis [47] :
-
Primary tumor thickness (Breslow depth)
-
Histologic ulceration, the presence of which significantly reduces survival at each tumor stage, even when regional lymph nodes are involved
-
Regional lymph node involvement, whether clinically evident or determined by sentinel lymph node biopsy and/or in-transit disease (lymphatic involvement)
-
Dermal mitotic rate in the primary tumor, which is not included in current melanoma staging but is an important prognostic factor across all tumor categories
Cutaneous melanoma (stages I and II) prognosis
Overall survival for patients with metastatic melanoma has improved dramatically since 2011, when the US Food and Drug Administration approved the first immune checkpoint inhibitor (ipilimumab) and targeted therapy (vemurafenib). Melanoma-specific survival data from the international collaborative database for the American Joint Committee on Cancer 8th edition (AJCC-8) does not incorporate contemporary melanoma outcomes data and so is likely underestimated. [47]
Thin [T1] primaries (≤1 mm) are associated with a 5-year survival rate of 99% and 10-year survival of 96-98% depending on the presence or absence of histologic ulceration and/or thickness < 0.8 mm vs ≥0.8 mm, per AJCC-8 melanoma staging.
Intermediate-thickness melanomas (1.01-4 mm) are associated with 5-year survival rate of 86-94%, depending on thickness (> 1.0-2 mm [T2], > 2.0-4 mm [T3]) and presence or absence of ulceration of the primary tumor, and negative sentinel lymph node biopsy status.
Patients with the thickest tumors (> 4 mm) have a 5-year survival rate of 90% without ulceration, compared with 82% with an ulcerated primary, in the absence of sentinel lymph node involvement. As noted, dermal mitotic rate (number of mitosis per mm2) may significantly impact prognosis, with mitotic rate 11/mm2 reducing 5-year survival to 84% (regardless of tumor thickness) [47]
Stage III disease prognosis
Regional lymph node metastasis is heterogeneous, with 5-year survival rates ranging from 32-93%, [47] depending on the number of nodes involved, microscopic or macroscopic (matted nodes/gross extracapsular extension) disease, and tumor thickness/ulceration status of the primary melanoma.
Lymphatic metastasis is associated with 68-75% 5-year survival rates, depending on whether microscopic and/or macroscopic disease is evident. Survival rates are worse in the setting of concomitant regional nodal metastasis.
Neoadjuvant approaches to bulky, resectable stage III disease are now considered standard, with initial systemic immunotherapy followed by surgery to assess for pathologic response of nodal or in-transit metastasis.
Stage IV disease prognosis
Prior to the advent of immune checkpoint inhibitors (ie, ipilimumab, nivolumab, pembrolizumab) and targeted therapy (ie, BRAF/MEK inhibitors) for melanoma, prognosis for distant metastatic disease was extremely poor, with median survival of only 6-9 months and 5-year survival rates of less than 20%, depending on the site(s) of metastasis.
With immune checkpoint blockade or targeted therapy, high overall response rates and disease-free survival has become the norm in patients with unresectable stage III and IV melanoma. Durable, complete responses have been observed with immune checkpoint inhibitors, which are also associated with improved relapse-free survival in the adjuvant setting for resected stage IIB/C, III, and IV melanoma. [48, 49]
Mortality/morbidity
While melanoma accounts for roughly 4% of all skin cancers, it is responsible for over 62% of skin cancer deaths. [1] Despite favorable trends in mortality for patients with advanced disease, treatment of melanoma in its early stages provides the best opportunity for cure.
US mortality/morbidity
An estimated 8430 melanoma-related deaths are expected in 2025 (5470 men and 2960 women). [1] From 2013 to 2016, overall mortality decreased by 17.9% (annual percent change [APC] -6.2%), with the most notable declines among men 50 years and older (APC -8.3%) observed since 2014, [2] following the advent of the modern therapeutic era. However, there remains a disproportionate burden of melanoma deaths among older White men and individuals of lower socioeconomic status across all racial and ethnic groups.
Worldwide mortality/morbidity
Cutaneous melanoma survival rates are higher in developed countries (ie, 94% 5-year relative survival in the US [41] ) than in countries with lower national levels of economic development. [39] Increased educational efforts regarding clinical warning signs and improved healthcare access in developed areas are associated with earlier diagnosis and treatment, with potential cure of thinner lesions and improved access to life-saving therapies for individuals with advanced disease.

Patient Education
Educate patients with a history of melanoma regarding the following:
-
Sun-protective measures (including sun-protective clothing and use of sunscreens)
-
Skin self-examinations for new primary melanoma, particularly important in individuals with numerous moles (common or atypical) and/or a strong family history of melanoma
-
Possible recurrence within the melanoma scar (visible pigmentation and/or nodularity in and around the excision scar
-
Screening of first-degree relatives, particularly if they have a history of atypical moles
-
Potential referral to a cancer genetics counselor for individuals with 2 or more invasive melanomas (personal or on the same side of the family) or multiple “cancer events” in the patient or family, including invasive melanoma(s) and pancreatic cancer, for discussion of germline genetic testing for the CDKN2A (P16) mutation or other pathogenic variants related to cancer syndromes. [50] However, a negative genetic testing result does not affect the need for ongoing dermatologic surveillance in patients at increased risk or with a history of multiple primary melanomas.

-
Superficial spreading melanoma, left breast, 1.3-mm Breslow depth.
-
Lentigo maligna melanoma, right lower cheek. Centrally located erythematous papule represents invasive melanoma with surrounding macular lentigo maligna (melanoma in situ).
-
Acral lentiginous melanoma (1-mm Breslow depth), left sole. Diagnostic punch biopsy site is located superiorly.
-
Malignant melanoma. Courtesy of Hon Pak, MD.
-
Cutaneous melanoma with characteristic asymmetry, irregular borders, and color variation. Courtesy of Wendy Brick, MD.