
Request Quote
| Product | Product Code | SAFETY DATA | Technical data | |
|---|---|---|---|---|
(2N) 99% Silver Nitrate |
AG-NAT-02 | SDS > | Data Sheet > | |
(3N) 99.9% Silver Nitrate |
AG-NAT-03 | SDS > | Data Sheet > | |
(4N) 99.99% Silver Nitrate |
AG-NAT-04 | SDS > | Data Sheet > | |
(5N) 99.999% Silver Nitrate |
AG-NAT-05 | SDS > | Data Sheet > | |
(6N) 99.9999% Silver Nitrate |
AG-NAT-06 | SDS > | Data Sheet > |

 Silver Nitrate is a highly water soluble crystalline Silver source for uses compatible with nitrates and lower (acidic) pH. All metallic nitrates are inorganic salts of a given metal cation and the nitrate anion. The nitrate anion is a univalent (-1 charge) polyatomic ion composed of a single nitrogen atom ionically bound to three oxygen atoms (Formula: NO3) for a total formula weight of 62.05. Nitrate compounds are generally soluble in water. Nitrate materials are also oxidizing agents. When mixed with hydrocarbons, nitrate compounds can form a flammable mixture. Nitrates are excellent precursors for production of ultra high purity compounds and certain catalyst and nanoscale (nanoparticles and nanopowders) materials. Silver Nitrate is generally immediately available in most volumes. High purity, submicron and nanopowder forms may be considered. American Elements produces to many standard grades when applicable, including Mil Spec (military grade); ACS, Reagent and Technical Grade; Food, Agricultural and Pharmaceutical Grade; Optical Grade, USP and EP/BP (European Pharmacopoeia/British Pharmacopoeia) and follows applicable ASTM testing standards. Typical and custom packaging is available. Additional technical, research and safety (MSDS) information is available as is a Reference Calculator for converting relevant units of measurement.
Silver Nitrate is a highly water soluble crystalline Silver source for uses compatible with nitrates and lower (acidic) pH. All metallic nitrates are inorganic salts of a given metal cation and the nitrate anion. The nitrate anion is a univalent (-1 charge) polyatomic ion composed of a single nitrogen atom ionically bound to three oxygen atoms (Formula: NO3) for a total formula weight of 62.05. Nitrate compounds are generally soluble in water. Nitrate materials are also oxidizing agents. When mixed with hydrocarbons, nitrate compounds can form a flammable mixture. Nitrates are excellent precursors for production of ultra high purity compounds and certain catalyst and nanoscale (nanoparticles and nanopowders) materials. Silver Nitrate is generally immediately available in most volumes. High purity, submicron and nanopowder forms may be considered. American Elements produces to many standard grades when applicable, including Mil Spec (military grade); ACS, Reagent and Technical Grade; Food, Agricultural and Pharmaceutical Grade; Optical Grade, USP and EP/BP (European Pharmacopoeia/British Pharmacopoeia) and follows applicable ASTM testing standards. Typical and custom packaging is available. Additional technical, research and safety (MSDS) information is available as is a Reference Calculator for converting relevant units of measurement. | Compound Formula | AgNO3 |
|---|---|
| Molecular Weight | 169.87 |
| Appearance | Colorless to white crystals or powder |
| Melting Point | 212 °C |
| Boiling Point | 440 °C |
| Density | 5.35 g/cm3 |
| Solubility in H2O | N/A |
| Exact Mass | 168.893 g/mol |
| Monoisotopic Mass | 168.892914 Da |
| Signal Word | Danger |
|---|---|
| Hazard Statements | H272-H314-H410 |
| Hazard Codes | C,N |
| Risk Codes | 34-50/53 |
| Safety Statements | 26-45-60-61 |
| RTECS Number | VW4725000 |
| Transport Information | UN 1493 5.1/PG 2 |
| WGK Germany | 3 |
| GHS Pictogram |
Image
Image
|
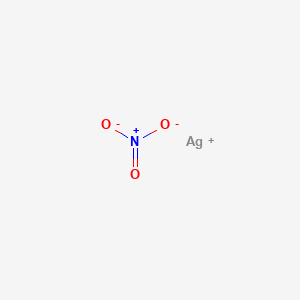
| Linear Formula | AgNO3 |
|---|---|
| Pubchem CID | 24470 |
| MDL Number | MFCD00003414 |
| EC No. | 231-853-9 |
| IUPAC Name | silver; nitrate |
| Beilstein/Reaxys No. | N/A |
| SMILES | [N+](=O)([O-])[O-].[Ag+] |
| InchI Identifier | InChI=1S/Ag.NO3/c;2-1(3)4/q+1;-1 |
| InchI Key | SQGYOTSLMSWVJD-UHFFFAOYSA-N |
| Chemical Formula | |
| Molecular Weight | |
| Standard InchI | |
| Appearance | |
| Melting Point | |
| Boiling Point | |
| Density |
Customer Reviews
Customers For Silver Nitrate Have Also Viewed
Related Applications, Forms & Industries for Silver Nitrate

Each business day American Elements' scientists & engineers post their choice for the most exciting materials science news of the day
See more Silver products. Silver (atomic symbol: Ag, atomic number: 47) is a Block D, Group 11, Period 5 element with an atomic weight of 107.8682.  The number of electrons in each of Silver's shells is 2, 8, 18, 18, 1 and its electron configuration is [Kr]4d10 5s1. The silver atom has a radius of 144 pm and a Van der Waals radius of 203 pm. Silver was first discovered by Early Man prior to 5000 BC. In its elemental form, silver has a brilliant white metallic luster.
The number of electrons in each of Silver's shells is 2, 8, 18, 18, 1 and its electron configuration is [Kr]4d10 5s1. The silver atom has a radius of 144 pm and a Van der Waals radius of 203 pm. Silver was first discovered by Early Man prior to 5000 BC. In its elemental form, silver has a brilliant white metallic luster.  It is a little harder than gold and is very ductile and malleable, being exceeded only by gold and perhaps palladium. Pure silver has the highest electrical and thermal conductivity of all metals and possesses the lowest contact resistance. It is stable in pure air and water, but tarnishes when exposed to ozone, hydrogen sulfide, or air containing sulfur. It is found in copper, copper-nickel, lead, and lead-zinc ores, among others. Silver was named after the Anglo-Saxon word "seolfor" or "siolfur," meaning 'silver'.
It is a little harder than gold and is very ductile and malleable, being exceeded only by gold and perhaps palladium. Pure silver has the highest electrical and thermal conductivity of all metals and possesses the lowest contact resistance. It is stable in pure air and water, but tarnishes when exposed to ozone, hydrogen sulfide, or air containing sulfur. It is found in copper, copper-nickel, lead, and lead-zinc ores, among others. Silver was named after the Anglo-Saxon word "seolfor" or "siolfur," meaning 'silver'.
See more Nitrogen products. Nitrogen is a Block P, Group 15, Period 2 element. Its electron configuration is [He]2s22p3. Nitrogen is an odorless, tasteless, colorless and mostly inert gas. It is the seventh most abundant element in the universe and it constitutes 78.09% (by volume) of Earth's atmosphere. Nitrogen was discovered by Daniel Rutherford in 1772.




